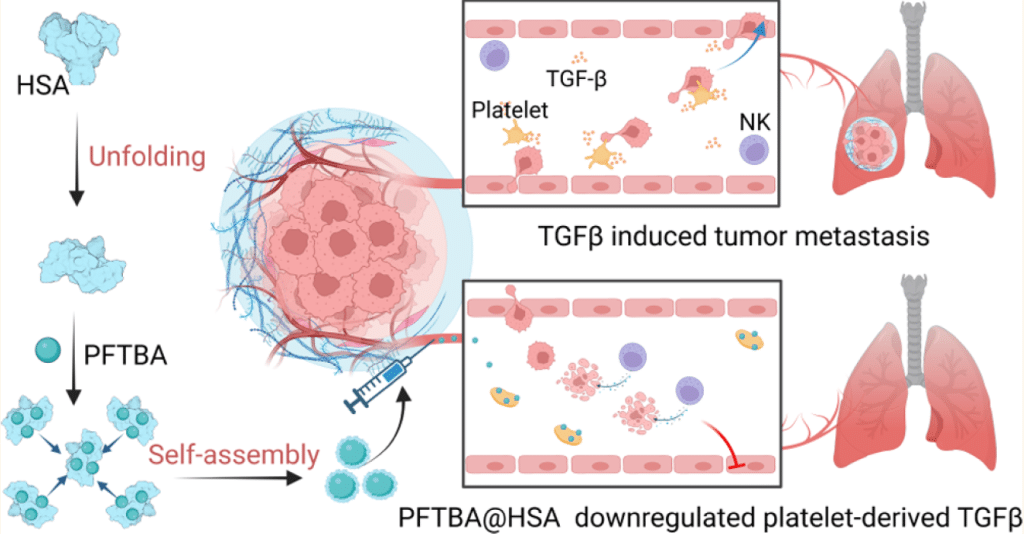
Tumor metastasis contributes to the low overall survival of tumor patients, while transforming growth factor-β (TGFβ) has been recognized as a prominently promoting factor in the development of tumor metastasis. Platelets reserve abundant TGFβ, which will be secreted to peripheral blood after activation, and they are the dominant source of circulating TGFβ. Therefore, downregulation of platelet-derived TGFβ is expected to inhibit the metastasis of circulating tumor cells. Here, unfolded human serum albumin (HSA)-coated perfluorotributylamine (PFTBA) nanoparticles were constructed to display a favorable platelet delivery and an antiplatelet effect to downregulate platelet-derived TGFβ in vitro and in blood plasma. PFTBA@HSA-mediated TGFβ downregulation impaired epithelial-mesenchymal transition of tumor cells as well as their migration and invasion behaviors and enhanced immune surveillance of NK cells. Intravenous injection of PFTBA@HSA effectively reduced tumor metastasis on the lungs or liver to improve the survival rate of mice on multiple metastatic models, including CT26 colon cancer, B16F10 melanoma, and 4T1 breast cancer. Compared with the clinical antiplatelet drug ticagrelor, PFTBA@HSA reduced bleeding risk when displaying a favorable downregulation on platelet-derived TGFβ, thereby obtaining a higher therapy benefit. Together, this study confirmed that downregulation of platelet-derived TGFβ by PFTBA@HSA will be a potential approach and therapeutic candidate for the prevention of tumor metastasis.
2023-05-07


